Reviewer Summary
CMS-TM1 in One Pass
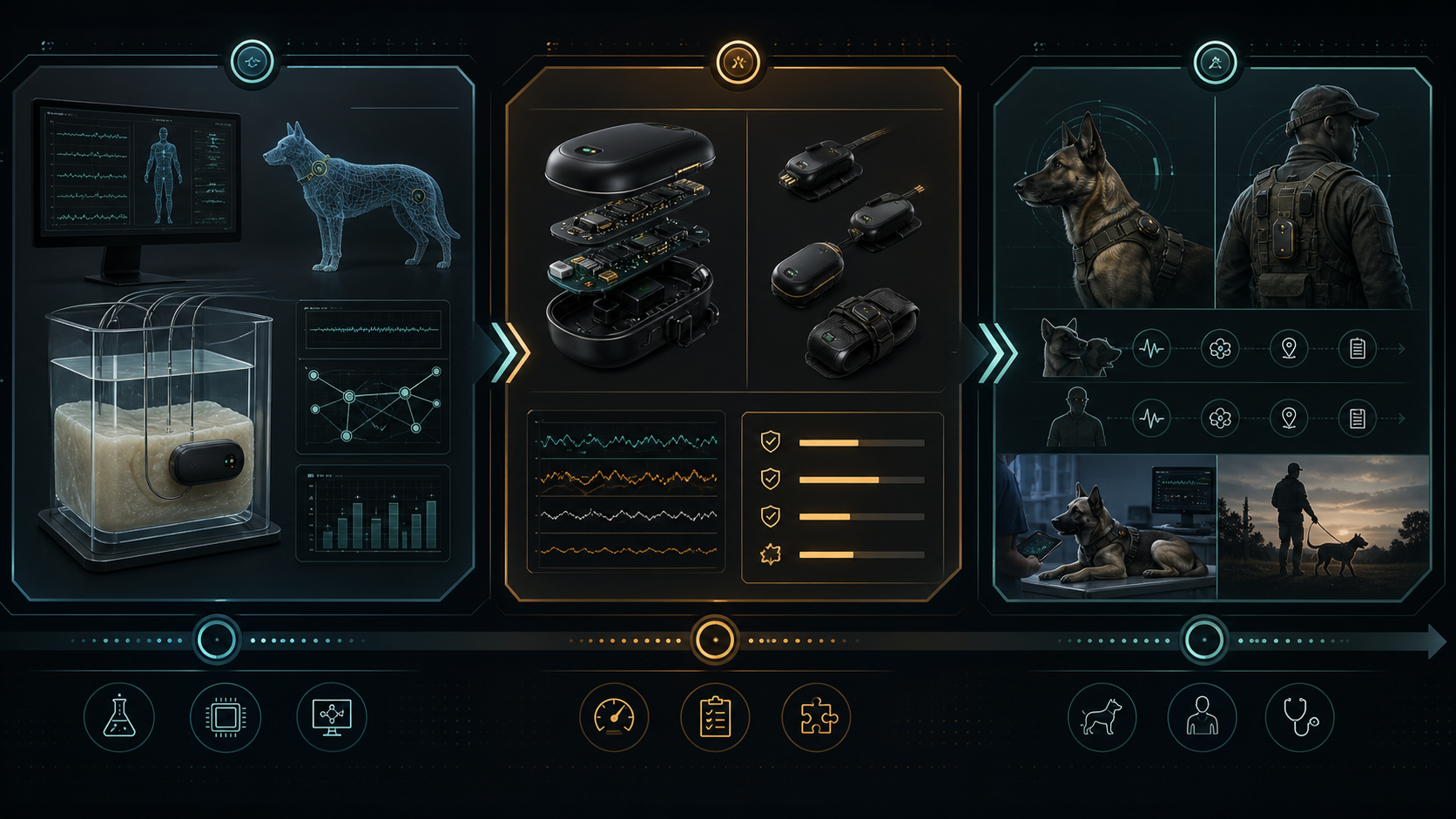
CMS-TM1 is a research-stage non-optical physiological status monitoring concept for military working dogs and human warfighters. The system uses a shared low-SWaP sensing pod, coherent radar, IMU context, edge DSP, and confidence scoring to support field triage and physiological status monitoring where optical and contact-based sensors are degraded by fur, motion, moisture, clothing, and unstable contact.
Phase I will not involve human or animal subjects. Feasibility will be evaluated through mechanical phantoms, synthetic fur/media layers, motion artifact injection, and in-silico physiological displacement models. The primary Phase I endpoint is respiration-related motion extraction and signal-quality confidence scoring. Cardiac-band micro-motion is treated as a secondary feasibility characterization endpoint.
Phase I validation: in vitro and in silico only. No human or animal subjects are proposed for Phase I.